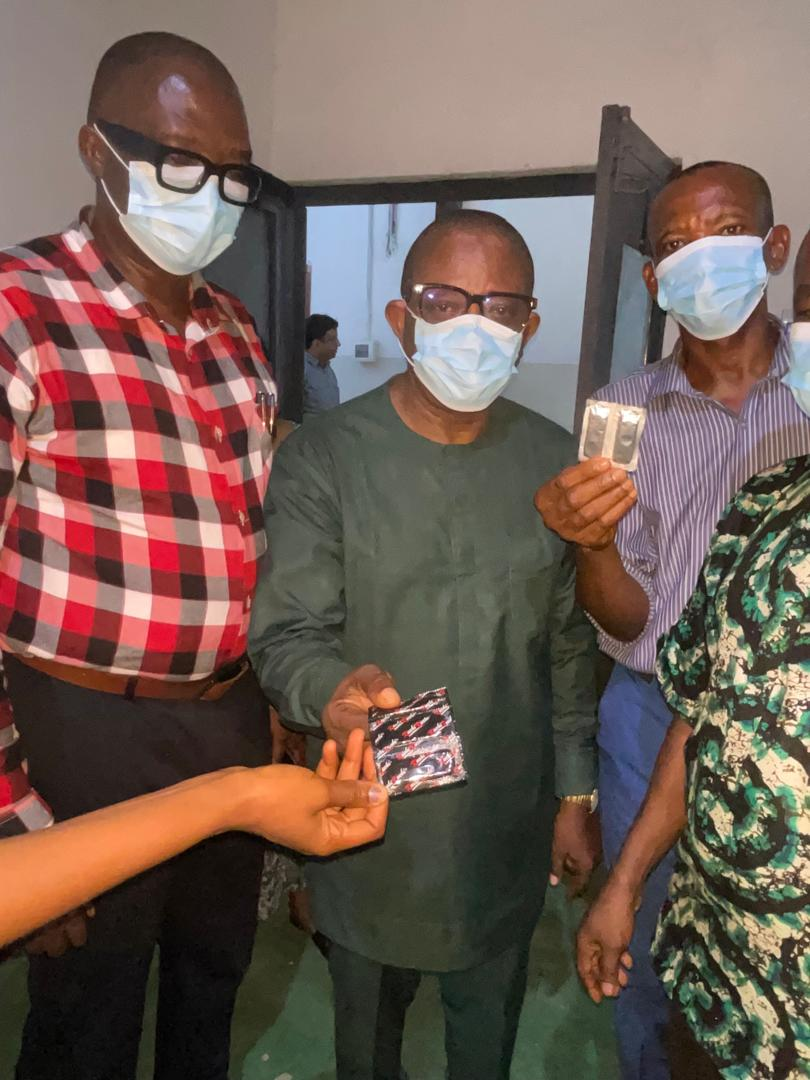
In a strategic push to achieve zero new HIV infections by 2030, the Cross River State Agency for the Control of AIDS (CR-SACA) has conducted an assessment of Agrim Pharmaceutical and Health Care FZE, a condom manufacturing facility located within the Calabar Export Processing Zone (EPZ).
The initiative aims to bolster local production and slash Nigeria’s heavy reliance on foreign imports with current data suggesting that Nigeria spends approximately $18 million annually on condom imports, an expenditure that could be significantly reduced through domestic manufacturing.
Agrim Pharmaceutical is reportedly the first condom manufacturing facility in Nigeria and only the second of its kind in Africa. Its operations are poised to not only meet local demand but also serve as a catalyst for regional employment.
Speaking during the facility tour in Calabar, Thursday, the Director General of CR-SACA, Dr. Charles Iwara, emphasized that engaging the private sector is vital for securing access to preventive health commodities.
According to him, "This engagement is necessary to verify the authenticity and capacity of local manufacturers. We must ensure any collaboration aligns with stringent quality assurance standards and our overarching public health objectives."
The Director General noted that the visit was prompted by discussions at a recent national conference in Abuja regarding condom scarcity and the need for indigenous production.
Dr. Iwara explained that he refrained from making "categorical comments" at the summit until he could personally verify the facility's operational standards, stressing that a successful HIV response cannot happen in a vacuum.
"Coordinated partnerships are essential to expanding prevention coverage across the State," he added, highlighting the need for better visibility and supply chain coordination.
Dr. Daniels commended CR-SACA’s proactive approach, noting that stronger synergy between the government and private manufacturers is the fastest route to improving public health outcomes.
Earlier, Dr. Obo Obo Daniels, the company’s representative, who took the CR-SACA team on inspection of the production and packaging lines, quality control systems, and the overall operational setup, thanked the DG for contemplating collaboration with the company.
He informed the DG that the company was fully approved by the National Agency for Food and Drug Administration and Control (NAFDAC), adding that When fully active, the facility operates on a 24-hour shift system with a capacity to produce over 300,000 packs daily.
Dr Daniels further disclosed that the plant produces condoms in various variants, all adhering to international safety standards.
CR-SACA continues to work alongside international partners, such as the United Nations Population Fund (UNFPA), to promote safe sex practices. By integrating locally produced commodities into their distribution network, the agency hopes to ensure a more sustainable and reliable supply of HIV prevention tools for high-risk groups across Cross River State.
Asari Eso